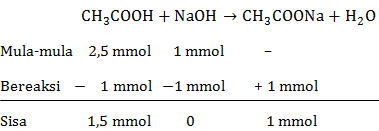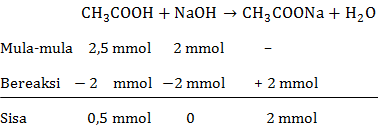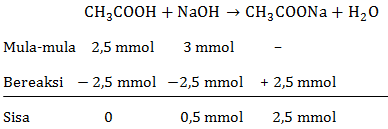Iklan
Pertanyaan
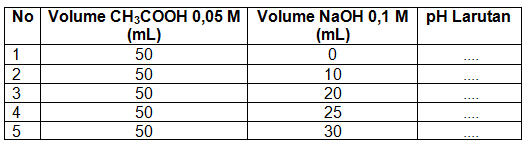
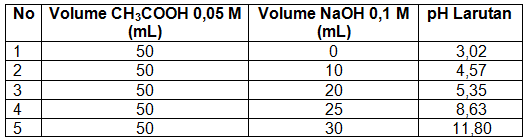
Membuat kurva titrasi 50 mL CH 3 COOH 0,05 M dengan NaOH 0,1 M. K a CH 3 COOH = 1 , 8 × 1 0 − 5 Gunakan tabel berikut untuk menentukan titik dalam kurva.
Membuat kurva titrasi 50 mL CH3COOH 0,05 M dengan NaOH 0,1 M.
Ka CH3COOH =
Gunakan tabel berikut untuk menentukan titik dalam kurva.
Iklan
SO
S. Oktasari
Master Teacher
Mahasiswa/Alumni Universitas Padjadjaran
Jawaban terverifikasi
1
0.0 (0 rating)
Iklan
Pertanyaan serupa
RUANGGURU HQ
Jl. Dr. Saharjo No.161, Manggarai Selatan, Tebet, Kota Jakarta Selatan, Daerah Khusus Ibukota Jakarta 12860
Produk Ruangguru
Bantuan & Panduan
Hubungi Kami
©2026 Ruangguru. All Rights Reserved PT. Ruang Raya Indonesia