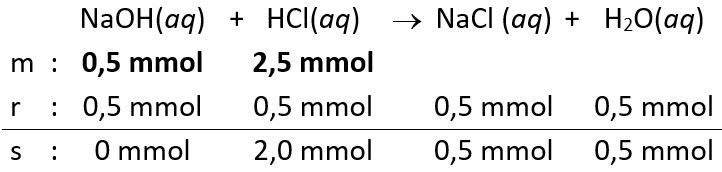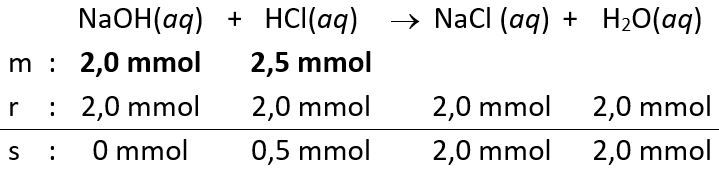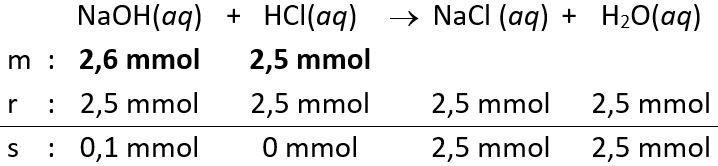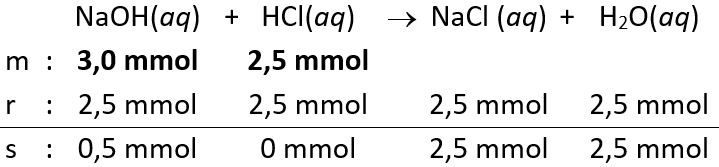Iklan
Pertanyaan
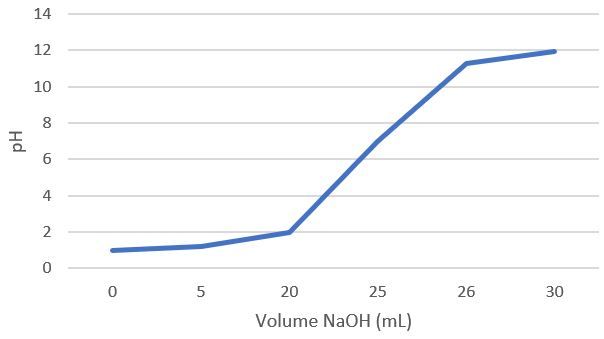
Sebanyak 25 mL larutan HCl dititrasi dengan larutan NaOH 0,1 M menggunakan indikator BTB. Setelah tercapai titik ekuivalen yang ditandai timbulnya warna biru, memerlukan larutan NaOH sebanyak 25 mL. Hitunglah konsentrasi molaritas ! Hitung pH larutan pada saat:Volume belum dicampurkan atau diteteskan. Volume yang dicampurkan atau diteteskan sebanyak 5 mL. Volume yang dicampurkan atau diteteskan sebanyak 20mL Volume yang dicampurkan atau diteteskan sebanyak 26mL Volume yang dicampurkan atau diteteskan sebanyak 30mL Buatlah kurva titrasi volume dengan pH dari perhitungan 2.1, 2.2, 2.3, 2.4, dan 2.5.
Sebanyak 25 mL larutan dititrasi dengan larutan NaOH 0,1 M menggunakan indikator BTB. Setelah tercapai titik ekuivalen yang ditandai timbulnya warna biru, memerlukan larutan sebanyak 25 mL.
- Hitunglah konsentrasi molaritas
!
- Hitung pH larutan pada saat:Volume
belum dicampurkan atau diteteskan.
- Volume
yang dicampurkan atau diteteskan sebanyak 5 mL.
- Volume
yang dicampurkan atau diteteskan sebanyak 20 mL
- Volume
yang dicampurkan atau diteteskan sebanyak 26 mL
- Volume
yang dicampurkan atau diteteskan sebanyak 30 mL
- Volume
- Buatlah kurva titrasi volume
dengan pH dari perhitungan 2.1, 2.2, 2.3, 2.4, dan 2.5.
Iklan
SH
S. Hidayati
Master Teacher
Mahasiswa/Alumni Universitas Indonesia
Jawaban terverifikasi
6
3.0 (2 rating)
Iklan
Pertanyaan serupa
RUANGGURU HQ
Jl. Dr. Saharjo No.161, Manggarai Selatan, Tebet, Kota Jakarta Selatan, Daerah Khusus Ibukota Jakarta 12860
Produk Ruangguru
Bantuan & Panduan
Hubungi Kami
©2026 Ruangguru. All Rights Reserved PT. Ruang Raya Indonesia