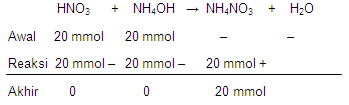Iklan
Iklan
Pertanyaan
Sebanyak 100 mLlarutan HNO 3 0,2 Mdireaksikan dengan 100 mL NH 4 OH 0,2 M.Jika garam yang dihasilkan diencerkan dengan 600 mL air, pH larutan garam setelahpengenceran adalah … ( K b NH 3 = 1 0 − 5 )
Sebanyak 100 mL larutan 0,2 M direaksikan dengan 100 mL 0,2 M. Jika garam yang dihasilkan diencerkan dengan 600 mL air, pH larutan garam setelah pengenceran adalah ()
2 - log 5
5 - log 6
5 - log 1
6 - log 5
6 - log 1
Iklan
SL
S. Lubis
Master Teacher
Mahasiswa/Alumni Universitas Sumatera Utara
Jawaban terverifikasi
170
4.4 (5 rating)
nt
nayla tiara rizky
ga bisa di akses
Iklan
Iklan
Pertanyaan serupa
RUANGGURU HQ
Jl. Dr. Saharjo No.161, Manggarai Selatan, Tebet, Kota Jakarta Selatan, Daerah Khusus Ibukota Jakarta 12860
Produk Ruangguru
Bantuan & Panduan
Hubungi Kami
©2024 Ruangguru. All Rights Reserved PT. Ruang Raya Indonesia