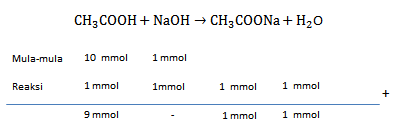Iklan
Pertanyaan
Larutan CH 3 COOH mempunyai pH=2 , jika diencerkan hingga 100 kali kemudian sebanyak 100 ml larutan tersebut direaksikan dengan 100 ml NaOH 0,01 M. Berapakah pH setelah pengenceran dan setelah penambahan ?
Larutan mempunyai pH=2 , jika diencerkan hingga 100 kali kemudian sebanyak 100 ml larutan tersebut direaksikan dengan 100 ml 0,01 M. Berapakah pH setelah pengenceran dan setelah penambahan ?
Iklan
NP
N. Puspita
Master Teacher
Jawaban terverifikasi
77
0.0 (0 rating)
Iklan
Pertanyaan serupa
RUANGGURU HQ
Jl. Dr. Saharjo No.161, Manggarai Selatan, Tebet, Kota Jakarta Selatan, Daerah Khusus Ibukota Jakarta 12860
Produk Ruangguru
Bantuan & Panduan
Hubungi Kami
©2025 Ruangguru. All Rights Reserved PT. Ruang Raya Indonesia