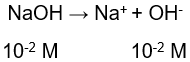Iklan
Pertanyaan
Tentukan massa NaOH yang mempunyai p H 12 dalam 250 mL larutan! ( A r Na = 23, O = 16, dan H = 1)
Tentukan massa NaOH yang mempunyai pH 12 dalam 250 mL larutan! ( Na = 23, O = 16, dan
H = 1)
20 gram
40 gram
80 gram
120 gram
160 gram
Iklan
SL
S. Lubis
Master Teacher
Jawaban terverifikasi
1
0.0 (0 rating)
Iklan
Pertanyaan serupa
RUANGGURU HQ
Jl. Dr. Saharjo No.161, Manggarai Selatan, Tebet, Kota Jakarta Selatan, Daerah Khusus Ibukota Jakarta 12860
Produk Ruangguru
Bantuan & Panduan
Hubungi Kami
©2026 Ruangguru. All Rights Reserved PT. Ruang Raya Indonesia