Iklan
Pertanyaan
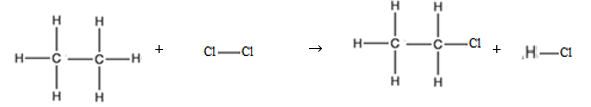
Data nilai energi ikatan rata-rata diketahui sebagai berikut C 2 H 6 ( g ) + C l 2 ( g ) → C 2 H 5 Cl ( g ) + H Cl ( g ) Nilai entalpi reaksi di atas adalah.....
Data nilai energi ikatan rata-rata diketahui sebagai berikut
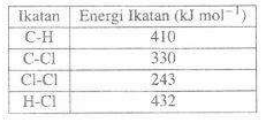
Nilai entalpi reaksi di atas adalah.....
Iklan
SL
S. Lubis
Master Teacher
Mahasiswa/Alumni Universitas Sumatera Utara
Jawaban terverifikasi
1
4.3 (6 rating)
R
RachelSiallgn
Pembahasan tidak menjawab soal
Iklan
Pertanyaan serupa
RUANGGURU HQ
Jl. Dr. Saharjo No.161, Manggarai Selatan, Tebet, Kota Jakarta Selatan, Daerah Khusus Ibukota Jakarta 12860
Produk Ruangguru
Bantuan & Panduan
Hubungi Kami
©2026 Ruangguru. All Rights Reserved PT. Ruang Raya Indonesia